Imagine struggling with constant nasal congestion, headaches, and loss of smell. For millions suffering from chronic rhinosinusitis, this is a daily reality.
On January 16, a groundbreaking study led by Professor Hai Qi's team from Tsinghua Medicine, Professor Luo Zhang's team from Beijing Tongren Hospital, Department of Otorhinolaryngology-Head and Neck Surgery, and Associate Professor Jianbin Wang's team from Tsinghua University School of Life Sciences was published online in Nature under the title GZMK-expressing CD8⁺ T cells promote recurrent airway inflammatory diseases. The study uncovers a novel subset of memory CD8+ T cells and reveals a new immunological mechanism behind the recurrent flare-ups of chronic rhinosinusitis with nasal polyps (CRSwNP), offering new insights for treating this often intractable disease.
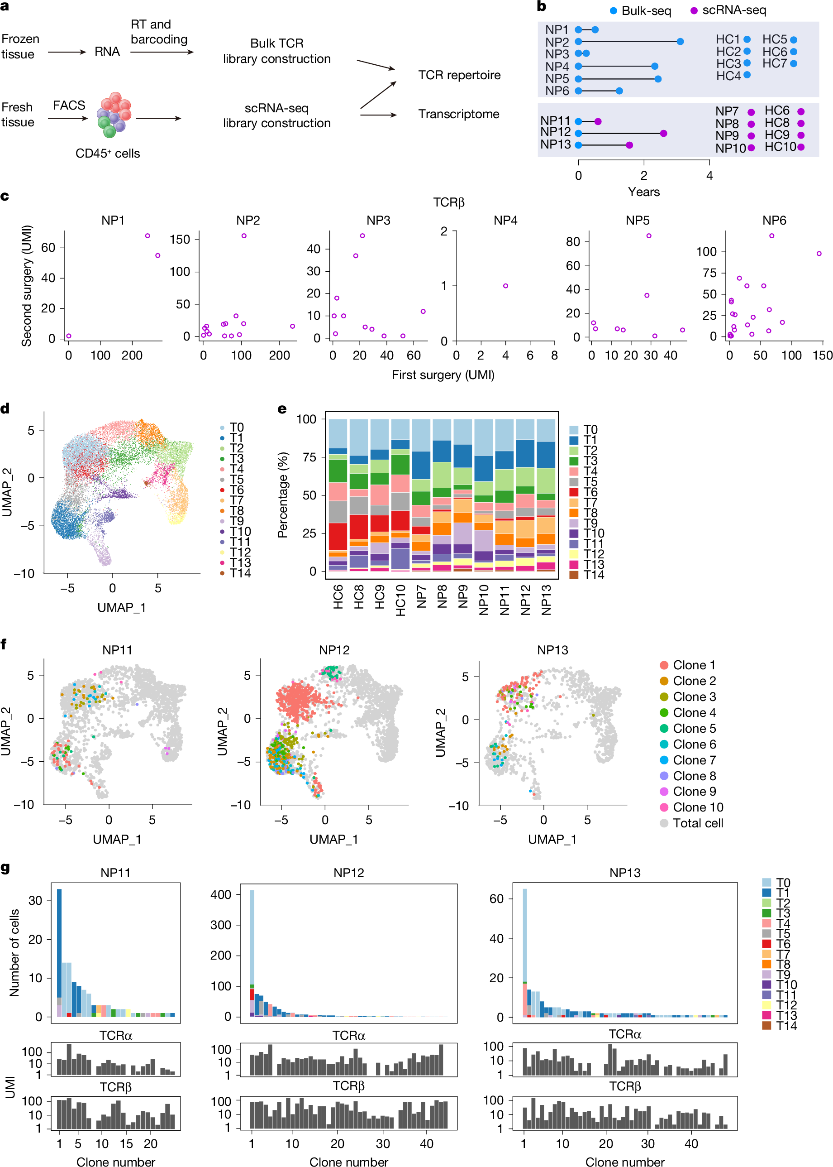
Fig. 1: Persisting T cell clones in recurrent NP.
Chronic rhinosinusitis is a common chronic nasal condition that affects hundreds of millions of people worldwide. While current treatments such as oral corticosteroids and nasal endoscopic sinus surgery, they have limited efficacy the 30+ million patients suffering from refractory CRSwNP, who often experience recurrence of symptoms. Research teams from Tsinghua University and Beijing Tongren Hospital have been working on this problem for almost a decade before ultimately locating the crux of the issue—a special subset of memory CD8+ T cells.
If white blood cells are akin to an army protecting human health, T lymphocytes are its “elite troops,” capable of highly targeted attacks. Among them, memory CD8+ T cells function like “special forces”: once they have been battle-tested against viruses, they can be rapidly mobilized when the same virus appears again. By using perforin, these cells punch holes in virus-infected cells and then inject granzyme B, a “toxin” that kills those cells, thereby destroying the virus. The body also uses a defensive mechanism called the complement system, typically activated by antibodies that recognize viruses. The complement system can pierce the surface of infected cells and destroy them, while also triggering inflammation by recruiting white blood cells from the bloodstream.
By analyzing nasal polyp tissue from patients, the research team has identified an unusual group of memory CD8+ T lymphocytes that secrete granzyme K—rather than the classic granzyme B—and appear in the polyp tissue each time the disease flares up. These T cells seem to recognize allergens and viruses that enter the airways. More importantly, granzyme K can directly activate the complement system without needing antibodies, thereby exacerbating tissue damage and amplifying inflammation. What makes matters worse is that these granzyme K–producing T cells are particularly “active,” continually migrating from peripheral blood into nasal tissues, causing flare-ups again and again, turning what might have been a minor disease into a persistent, hard-to-cure condition.
Building upon these critical discoveries, the researchers explored the potential of treating this disease by inhibiting granzyme K. Experiments in animal models revealed that removing or inhibiting granzyme K in CD8+ T lymphocytes significantly reduced inflammation and led to effective treatment outcomes. These results indicate that granzyme K and the memory CD8+ T cells that secrete it are key drivers of relapse in refractory chronic rhinosinusitis with nasal polyps—and could also serve as crucial biomarkers for forecasting disease progression and treatment responses.
By unveiling a new subset of memory CD8+ T cells, clarifying how granzyme K activates the complement system to boost inflammation, and presenting a new immunological mechanism behind chronic airway inflammation (as seen in chronic rhinosinusitis with nasal polyps), this study lays the groundwork for developing innovative treatments. Its publication signals that Chinese researchers have taken a leading position globally in investigating refractory chronic rhinosinusitis with nasal polyps and translating these findings into clinical practice.
Professor Qi Hai from Tsinghua University, Professor Zhang Luo from Beijing Tongren Hospital, Associate Professor Wang Jianbin from Tsinghua University, and Associate Researcher Liu Xin from Tsinghua Medicine are the corresponding authors of this paper. Research Fellow Lan Feng from Beijing Tongren Hospital, Ph.D. candidate Li Jizhou from the School of Life Sciences, Ph.D. candidate Miao Wenxuan from the School of Basic Medical Sciences, and Ph.D. candidate Sun Fei from the School of Life Sciences at Tsinghua University are the co-first authors of this paper. Professor Wang Chengshuo, Professor Wang Xiangdong, and Chief Physician Duan Su from Beijing Tongren Hospital made significant contributions to this project. The study was supported by the National Key R&D Program, Tsinghua University Independent Research Program, National Natural Science Foundation of China, the Shanxi Medical University–Tsinghua Medicine Frontier Biomedical Research Program, the National Health Commission, and the New Cornerstone Foundation.
Editor: Li Han

